Revolution Medicines, a biotech company, has been developing a new pancreatic cancer drug that showed strong results in a Phase 3 trial, nearly doubling how long patients lived. The company also said it cut the risk of death by about 60% compared to standard chemotherapy.
 The drug, called daraxonrasib, is a daily pill given to patients whose cancer had progressed after initial treatment.
The drug, called daraxonrasib, is a daily pill given to patients whose cancer had progressed after initial treatment.
In the study, people taking it lived an average of 13.2 months, compared to 6.7 months for those on chemotherapy – that’s an improvement of about six and a half months.
Mark Goldsmith, the CEO of Revolution Medicines, described the results as a major breakthrough and said the company is now focused on getting the drug to patients as quickly as possible.
He noted that outcomes like this haven’t been seen before in this type of trial for pancreatic cancer.
The company plans to apply for approval from the Food and Drug Administration soon, using a priority review program that could speed things up significantly.
 Pancreatic cancer is known for being especially aggressive, with one of the lowest survival rates among major cancers, at approximately 13% after five years.
Pancreatic cancer is known for being especially aggressive, with one of the lowest survival rates among major cancers, at approximately 13% after five years.
This new drug targets RAS mutations, which play a role in tumor growth and are found in most pancreatic cancer cases.
Doctors are hopeful with the results. Shubham Pant, a specialist at MD Anderson, said the improvement is a big deal compared to past studies, which often only extended survival by a short time.
He’s been involved with this drug’s development for years and said seeing these results has been emotional, especially when thinking about his patients.
The treatment has also been getting attention after former senator Ben Sasse shared his own experience with it. He said his tumors shrank significantly while taking the drug, though he also dealt with side effects like skin rash.
The company said rashes are a known side effect but are usually manageable.
 Doctors added that most patients do experience some form of rash, though more severe cases are less common; they can often be handled by adjusting treatment or using medications like antibiotics.
Doctors added that most patients do experience some form of rash, though more severe cases are less common; they can often be handled by adjusting treatment or using medications like antibiotics.
So far, the trial hasn’t raised any new safety concerns. The overall side effect profile was also considered manageable. More detailed results will be shared at an upcoming medical meeting.
Right now, the plan is to use the drug as a second option for patients whose cancer has already spread or gotten worse after trying another treatment.
There’s also another Phase 3 trial underway for people who are newly diagnosed.
Experts like Andrew Aguirre believe this drug could become a key building block for future treatments, especially when combined with other therapies.
People are starting to feel hopeful that targeting RAS could lead to better results, not just for pancreatic cancer, but for other hard to treat diseases too.
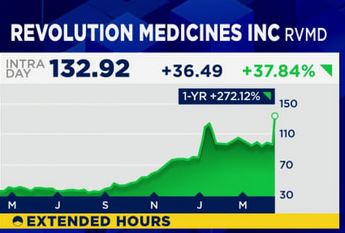
Since the announcement, shares of Revolution Medicines has jumped more than 30%.
The company is now valued at over $26 billion, though leadership says the main focus is on getting the drug approved and ready for patients as opposed to increasing profits.








